See What the Platform Produces
Real output from live deployments — structured intelligence generated automatically from raw trial data.
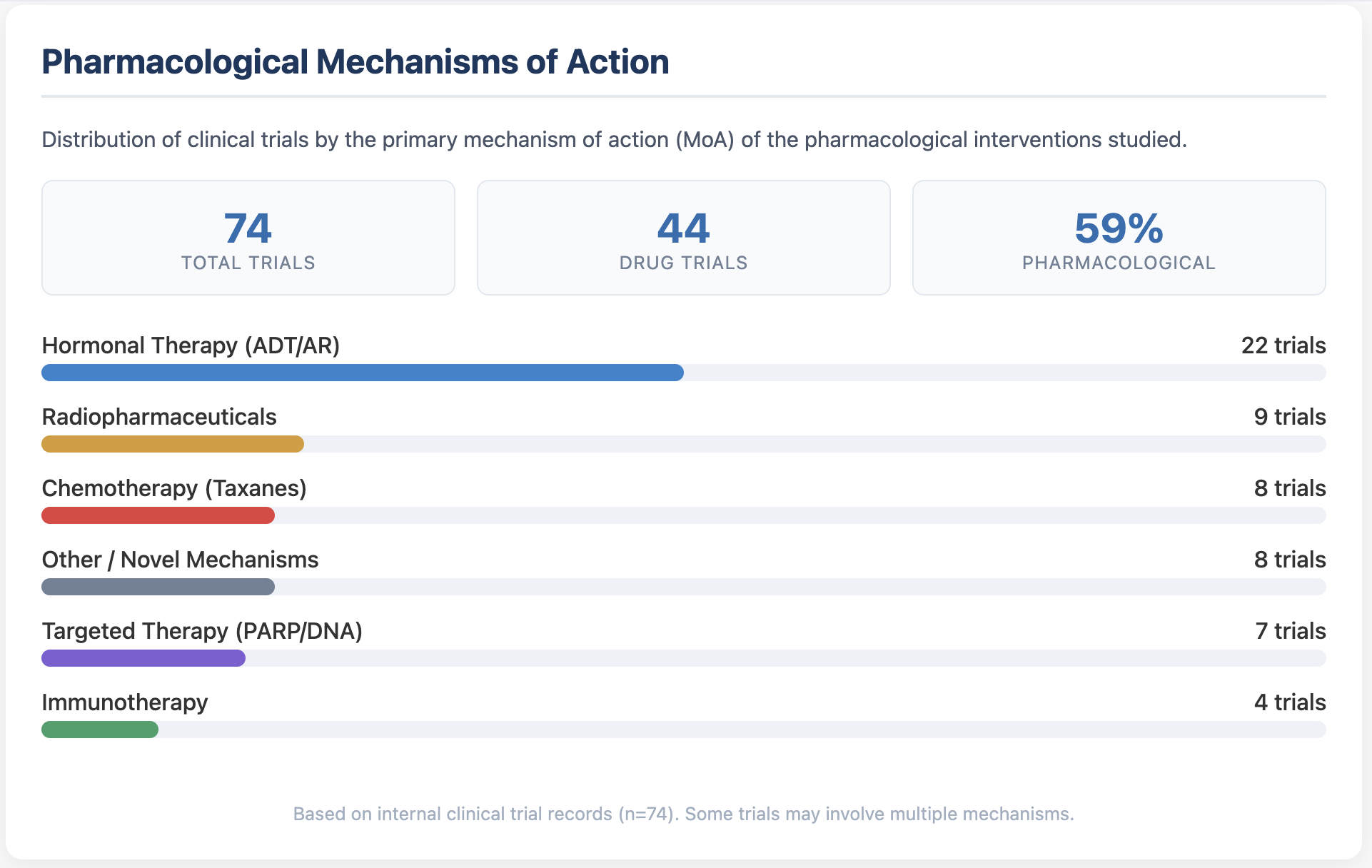
From Raw Protocols to Structured Insight
The platform automatically ingests trial documents and extracts the pharmacological mechanisms, drug classes, and intervention types — turning unstructured PDFs into queryable, structured knowledge.
- 100+ variables extracted per protocol
- Drug class and MoA classification
- Validated against clinical ontologies
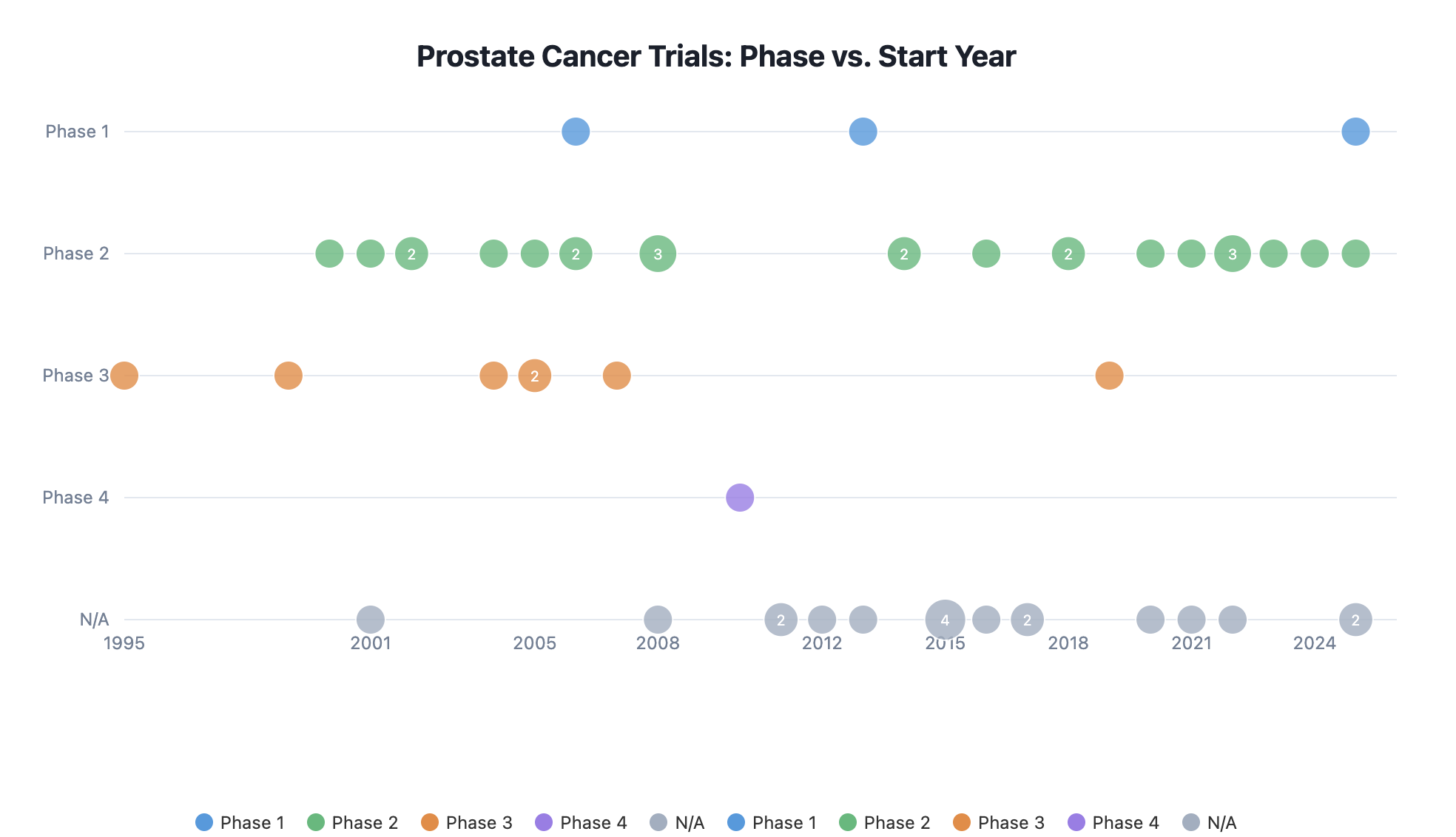
Decades of Trial History, Instantly Searchable
The platform maps every investigator's trial history across phases, years, and therapeutic areas — so site selection is based on proven track record, not guesswork or outdated spreadsheets.
- Phase and timeline visualisation per indication
- ClinicalTrials.gov + PubMed integrated
- Persistent profiles regardless of PI movement